 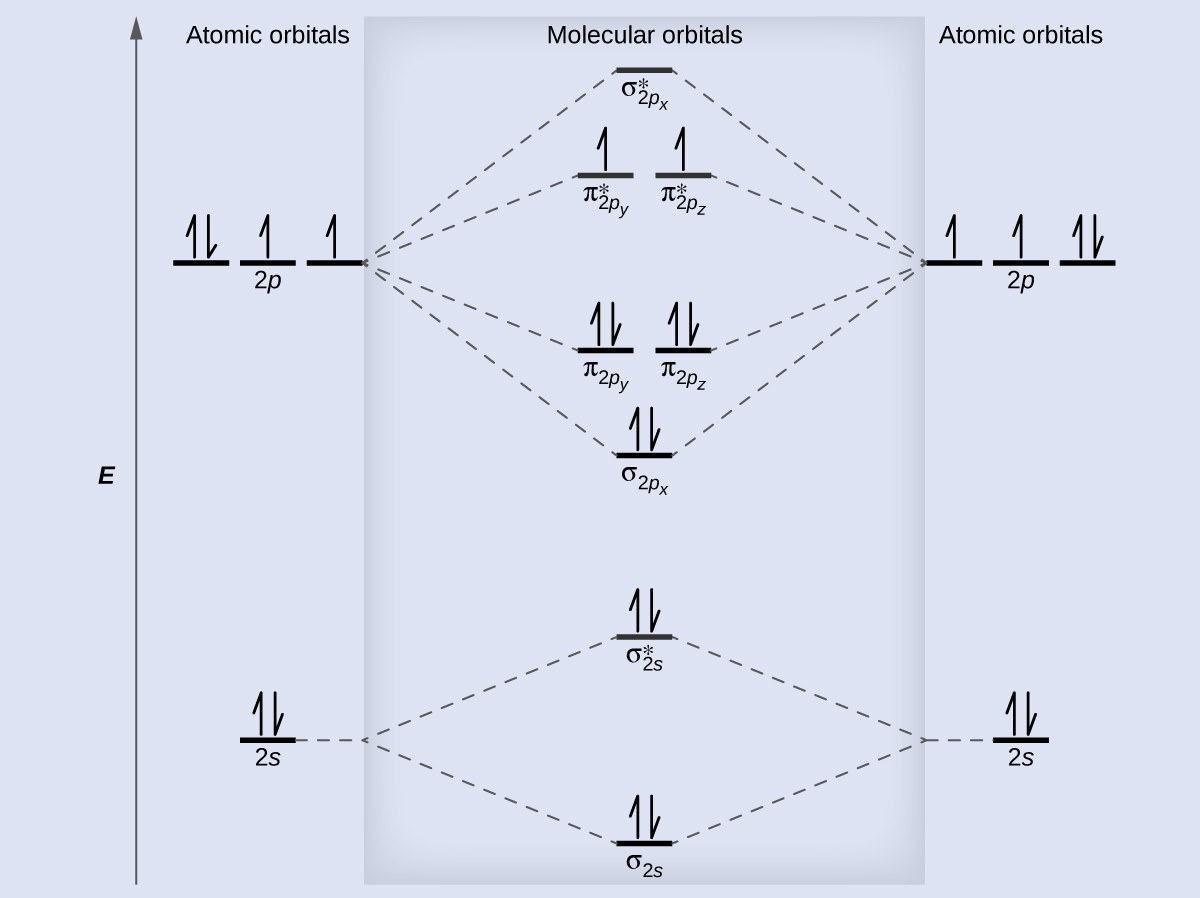
However, they are unequal hybrids, with more s in one and more p in the other.) We can picture sp hybrids making 4 combinations. (Basically they form sp hybrids, like you have seen before. And we can add some p-character to the s anti-bonding combination so that the overlap is worse, and it is less anti-bonding. This means that we can add some p-character to the bonding s combination, so the overlap is better. However, in elements with less difference between the 2s and 2p energies, like carbon, the 2s and 2p z orbitals can mix. Full valence MO diagram for F 2, showing all valence orbitals and electrons. You can see that including the 2s orbitals does not change the bond order, because both the bonding and anti-bonding combination are filled. So you get an MO diagram as shown in the figure. In molecules like F 2, they mostly interact separately, because the energy match between them is very bad. Do they do this separately? It turns out that sometimes they do and sometimes they don't. But we have to think more about the σ orbitals. The 2p orbitals that make π combinations don't do anything new. They are also smaller, so they have worse overlap with core orbitals on other atoms, and lower energy, so they have bad energy match with valence orbitals on other atoms. 
The core orbitals are completely full, so there can't be any net bonding between them. We don't have to worry about 1s orbitals, because their interactions don't have much effect on the properties of the molecule. Let's think about the orbitals we use to make MO diagrams for the first row elements, Li-Ne. Magnetic properties: Since each 2p x and 2p y MO contains unpaired electron, therefore B 2 molecule is paramagnetic.\) The two boron atom is B2 molecules are linked by one covalent bond. 6 in all, have to be accommodated in various molecular orbitals in the increasing order of their energies. In the formation of B 2 molecule, three valence electrons of each boron atom i.e. A number of valence electrons of each boron atom = 3. It is diamagnetic due to the absence of any unpaired electron.ī 2 molecule: The electronic configuration of B atom (Z = 5) isī 2 molecule is formed by the overlap of atomic orbitals of both boron atoms. Magnetic property: Since bond order is zero, Be 2 molecule does not exist. The molecular orbital electronic configuration, 4 in all, have to be accommodated in various molecular orbitals in the increasing order of their energies. Thus in the formation of Be 2 molecule, two outer electrons of each Be atom i.e. Number of valence electrons in Be atom = 2 (i) Be2 molecule: The electronic configuration of Be(Z = 4) is:īe 2 molecule is formed by the overlap of atomic orbitals of both beryllium atoms. (iv) Magnetic character: As it has two unpaired electrons one each in orbitals, it is paramagnetic. Since bond order is positive, it is quite stable. (c) ion: It is formed when molecule gains two electrons, (iv) Magnetic character Due to the presence of an unpaired electron in *2p x orbital, it is paramagnetic. Bond order being positive, N 2 is quite stable. This electron will go to either orbital of N 2 each of which is empty. (b) ion : It is formed by the gain of one electron by N 2 molecule. (iv) Magnetic character: Since 2p z orbital has one unpaired electron, therefore it is paramagnetic. (iii) Stability: As the bond order is positive, it is quite stable. This electron will be lost from 2p 7 orbital of nitrogen molecule. A) ion: It is formed from a molecule by the loss of one electron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
